Pulmonary immunity
INSERM U1019 – CNRS UMR9017 – Lille University – CHU Lille – Institut Pasteur de Lille

Presentation
Among respiratory diseases, allergic asthma and pulmonary infections with acute respiratory distress syndrome (SDRA) represent major public health issues. These diseases affect millions of people, are in constant increase and are a major cause of morbidity and mortality. These diseases share common key target cells involved in their pathogenesis: the endothelial cells as a barrier allowing the recruitment of inflammatory cells into the tissues, and the lymphoid cells, as major actors of the immune response to environmental challenges. Our goal is to evaluate how these cells and their mediators can orchestrate the host inflammatory reaction and tissue remodelling in response to allergens, microorganisms or stress, to characterize mechanisms of pathophysiology and to propose novel therapeutic strategies.
Highlights
- A dysregulation of trans-endothelial leukocyte diapedesis is a major factor involved in the severity of acute respiratory distress syndrome. Among the factors regulating the endothelial barrier, the endocan proteoglycan, through its binding to leukocyte LFA-1, inhibits trans-endothelial leukocyte migration. In contrast, the proteolytic cleavage fragment of endocan, p14, a biological competitor of endocan, inhibits its binding to LFA-1, restoring trans endothelial leukocyte adhesion and migration across the endothelium (Gaudet et al, J Leuk Biol, 2020).
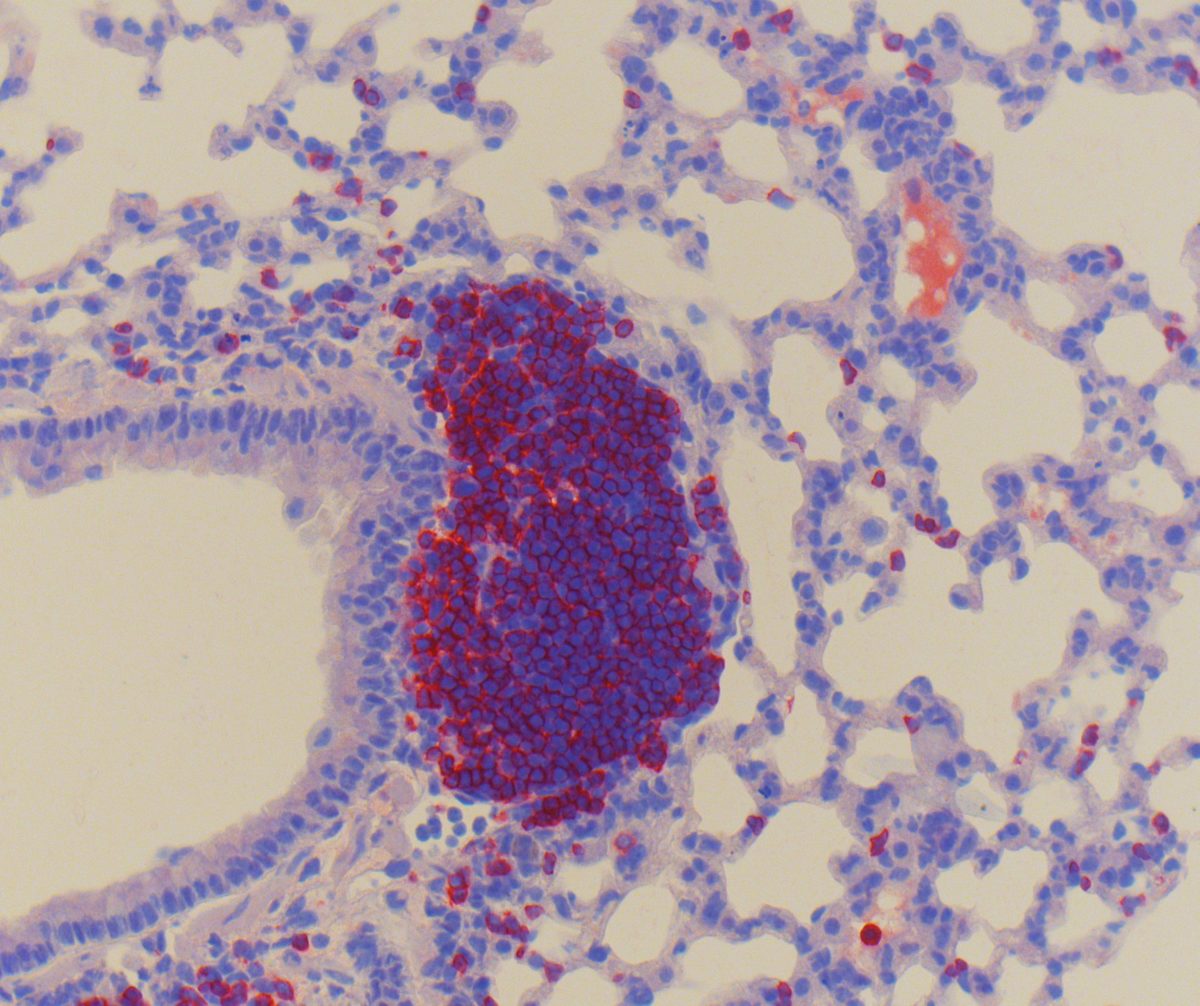
- Atmospheric pollution is deleterious in asthma. An exacerbating role of benzo-a-pyrene coated nanoparticles is observed in a model of house dust mite-induced asthma. This effect is linked to the recruitment of cell populations not induced in the asthma model at baseline, i.e. NKT-like cells, neutrophils and CD8+ T cells (Carrard et al, Allergy, 2020). Interestingly, house dust mite allergen, through its microbiota, is able to directly activate NOD1, a receptor recognizing Gram negative bacteria, resulting in the aggravation of asthma (PTR 18/16; ANR Innovasthma) (Ait Yahia et al, J Allergy Clin Immunol, 2021). Besides pollutants, viral infections by rhinoviruses are a major factor of asthma exacerbation. In this context, NK cells from severe asthma patients are less cytotoxic and produce less IFN-g in response to rhinovirus A9 than healthy subjects, and exhibit an exhausted phenotype, in relation with an increase of the check-point molecule Tim-3. This may participate in the aggravation of viro-induced exacerbations in these patients (Devulder et al, Eur Respir J, 2020). Moreover, in a model of neutrophilic asthma, inhibition of the transcription factor Aryl hydrocarbon receptor inhibits IL-22 production, neutrophil infiltration and airway hyperresponsiveness (Bouté et al, Allergy, 2021).
- Altogether, these data highlight new mechanisms involved in the severity of asthma and of acute respiratory distress syndrome, and open new therapeutic and diagnosis perspectives in these two diseases.
Transversal projects
 Circadian rhythm and lymphoid cells in asthma
Circadian rhythm and lymphoid cells in asthma
We have previously demonstrated that obesity exacerbates house dust mite-induced asthma by increasing lung Th and innate lymphoid cells. Obesity as well as asthma exhibit circadian variations, however, the underlying molecular mechanisms remain unclear. In collaboration with the team of Hélène Duez, the project evaluates if allergic asthma is associated with circadian variations of lymphoid cells , and if these modifications may impact the severity of asthma (CPER CTRL20 FEDER Circasthma).
 Vaccination using BPZE1 in house dust mite-induced asthma
Vaccination using BPZE1 in house dust mite-induced asthma
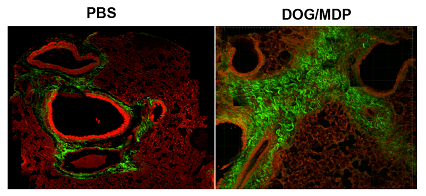
BPZE1 is a live attenuated vaccine of Bordetella pertussis, which has also non specific protective effects against infectious and inflammatory diseases, including asthma. In collaboration with the team of Nathalie Mielcarek/Camille Locht, the project analyzes if in a murine model of house dust mite-induced asthma, BPZE1 and different mutated forms can have a preventive/therapeutic effect.
Members
Anne TSICOPOULOS
DR2 Inserm, group leader
Céline CHENIVESSE
PU-PH, group leader
Philippe LASSALLE
CR1 Inserm
Patricia DE NADAI
MCF UDL
Daniel MATHIEU
PU-PH
Benoit WALLAERT
PU-PH, consultant
Thierry PEREZ
Hospital Practitioner
Nicolas JUST
Hospital Practitioner
Stéphanie FRY
Hospital Practitioner
Erika PARMENTIER
Hospital Practitioner
Anne PREVOTAT
Hospital Practitioner
Lidwine WEMEAU STERVINOU
Hospital Practitioner
Clément FOURNIER
Hospital Practitioner
Frédéric WALLYN
Hospital Practitioner
Alexandre GAUDET
CCA
Maxence HUREAU
CCA
Saliha AIT YAHIA
Engineer Inserm
Joanne BALSAMELLI
Technician IPL
Mélodie BOUTÉ
Post-doc
Daniel ALVAREZ-SIMON
Post-doc
Camille AUDOUSSET
PhD student
Publications
Ait Yahia S, Audousset C, Alvarez-Simon D, Vorng H, Togbe D, Marquillies P, Delacre M, Rose S, Bouscayrol H, Rifflet A, Quesniaux V, Gomperts Boneca I, Chamaillard M, Tsicopoulos A.
NOD1 sensing of house dust mite-derived microbiota promotes allergic experimental asthma.
J Allergy Clin Immunol. 2021, in press.
Carrard J, Marquillies P, Pichavant M, Visez N, Lanone S, Tsicopoulos A, Chenivesse C, Scherpereel A, de Nadaï P.
Chronic exposure to benzo(a)pyrene-coupled nanoparticles worsens inflammation in a mite-induced asthma mouse model.
Allergy. 2020 Oct 9. doi: 10.1111/all.14619. Online ahead of print.PMID: 33037642
Devulder J, Chenivesse C, Ledroit V, Fry S, Lobert PE, Hober D, Tsicopoulos A, Duez C.
Aberrant anti-viral response of natural killer cells in severe asthma.
Eur Respir J. 2020 May 14;55(5):1802422. doi: 10.1183/13993003.02422-2018. Print 2020 May.PMID: 32108047
Gaudet A, Portier L, Mathieu D, Hureau M, Tsicopoulos A, Lassalle P, De Freitas Caires N.
Cleaved endocan acts as a biologic competitor of endocan in the control of ICAM-1-dependent leukocyte diapedesis.
J Leukoc Biol. 2020 May;107(5):833-841. doi: 10.1002/JLB.3AB0320-612RR. Epub 2020 Apr 9.PMID: 32272492
Everaere L, Ait-Yahia S, Molendi-Coste O, Vorng H, Quemener S, LeVu P, Fleury S, Bouchaert E, Fan Y, Duez C, de Nadai P, Staels B, Dombrowicz D, Tsicopoulos A.
Innate lymphoid cells contribute to allergic airway disease exacerbation by obesity.
J Allergy Clin Immunol. 2016 Nov;138(5):1309-1318.e11. doi: 10.1016/j.jaci.2016.03.019. Epub 2016 Apr 4.PMID: 27177781
Keywords
Allergy ; Asthma ; Acute respiratory distress syndrome ; Endothelial cells ; Lymphoid cells immunity