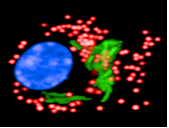
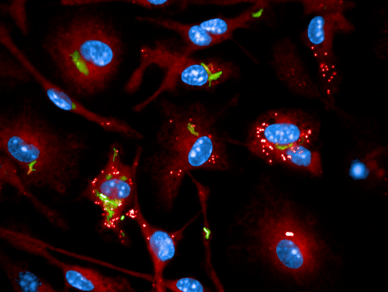
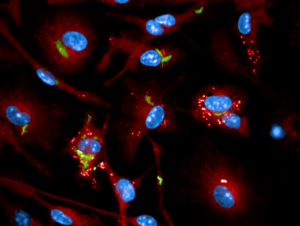
Chemogenomic of intracellular mycobacterium
INSERM U1019 – CNRS UMR9017 – Lille University – CHU Lille – Institut Pasteur de Lille
Presentation
The team projects focus on Tuberculosis (TB). TB is an infectious disease caused by Mycobacterium tuberculosis (Mtb) that results in millions of deaths annually worldwide and an increased incidence of reported drug-resistant cases. New drugs as well as novel drug targets are urgently needed. In order to realize radical advances in TB drug discovery, a better understanding of Mtb persistence and latency are necessary. In particular, this requires detailed elucidation of the mechanisms by which host cells control intracellular replication upon infection by pathogenic mycobacteria.
The team’s overall objective is to better understand TB pathogenesis to develop powerful strategies with enhanced efficacy, which allow the host to minimize or eradicate colonization by Mtb. The comprehensive picture of the molecular interactions between Mtb effectors and host cell proteins is necessary to generate the molecular tools for the development of antivirulence compounds that affect specific host pathways.
Highlights
- REDAMP project supported by Satt Nord (Société d’Accélération du Transfert de Technologie) aims to identify, improve and/or develop antimicrobial peptides (PAMs), derived from extremophiles animals as an alternative to antibiotics. Antibiotic resistance is a serious threat to global health, food security and development (World Health Organization WHO, 2016). Although all bacterial species are concerned by the acquisition of antibiotic resistance, some are prioritized for the identification and development of new molecules. One possible alternative to antibiotics is the development of antimicrobial peptides (MAPs), antibiotics produced naturally by all living organisms. PAMs from extremophile marine organisms, unlike those from terrestrial organisms (insects, amphibians, etc.), offer natural saline resistance but also tolerance to pH and temperature variations. They have been selected over thousands of years of evolution to be active, in extreme conditions, against proteobacteria, a family including all multi-resistant bacteria.
Transversal project
 Development of new potent analgesics
Development of new potent analgesics
The teams of Dr Priscille Brodin and Dr Laurent Marsollier, Research Directors at Inserm, have discovered an original molecular mechanism involved in pain control. Indeed, the researchers have shown that a lipid secreted by the bacterium, mycolactone, is responsible for the absence of pain by acting on the angiotensin 2 receptor 2 (AT2R) present at the level of the sensory neuron in the skin. Mycolactone then triggers a local discharge of cellular potassium via the TRAAK potassium channel, which prevents the transmission of nerve information responsibly for pain to the brain. The fact that a bacterial component activates a receptor to block a pain signal is particularly novel because most bacteria cause the opposite effect. This project is characterized by the development of a pain treatment, copying the natural effects of mycolactone. It has a long-lasting analgesic effect at very low doses and produces no side effects.
ANR Grant AT2R Traak Bionanalgesics coordinated by Priscille BRODIN (2018-2021).
Members
Priscille BRODIN
DR1 Inserm, group leader
ORCID number : 0000-0003-0991-7344
Aurélie TASIEMSKI
MCF Univ Lille
ORCID number : 0000-0003-3559-5115
François MASSOL
DR2 CNRS
ORCID number : 0000-0002-4098-955X
Arnaud MACHELART
CRCN Inserm
ORCID number : 0000-0002-4444-0011
Nathalie DEBOOSERE
IE IPL
ORCID number : 0000-0002-2873-5485
Alexandre VANDEPUTTE
IE Univ Lille
ORCID number : 0000-0003-2936-2411
Céline BOIDIN-WICHLACZ
IE CNRS
ORCID number : 0000-0002-0242-868X
Imène BELHAOUANE
PhD student
Tom BOURGUIGNON
PhD student
Ruben VAN DE WALLE
PhD student
Maxime CLENET
PhD student
Tâm LE MINH
PhD student
Publications
Paradoxical Roles of the MAL/Tirap Adaptor in Pathologies.
Frontiers in Immunology. 2020-09-25 DOI: 10.3389/fimmu.2020.569127. Part of ISSN: 1664-3224.
Multiplexed Quantitation of Intraphagocyte Mycobacterium tuberculosis Secreted Protein Effectors.
Cell Reports. 2018 DOI: 10.1016/j.celrep.2018.03.125. WOSUID: WOS:000432453100015.
Proteomics of Mycobacterium infection: Moving towards a Better Understanding of Pathogen-Driven immunomodulation.
Frontiers in Immunology 2018 DOI: 10.3389/fimmu.2018.00086. WOSUID: WOS:000423557200001.
Mycobacterium tuberculosis inhibits human innate immune responses via the production of TLR2 antagonist glycolipids.
Proceedings of the National Academy of Sciences. 2017. DOI: 10.1073/pnas.170784011.
Keywords
Tuberculosis (TB) ; Mycobacterium tuberculosis (Mtb) ; New drugs ; AT2R-Traak ; Tirap-CISH-vATPase ; Antimicrobial peptides ; Resistance development